Practice Problems Week 7
1. Find the oxidation number of chromium in each of the following compounds:
(a) ![]()
(b) 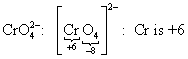
(c) ![]()
2. For each of the following equations, (a) complete and balance the equation; (b) determine if the reaction is redox or not; (c) for redox, write the oxidation and reduction half reactions and (d) indicate the element that is reduced, the element that is oxidized, the reducing agent and the oxidizing agent.
(i) sodium carbonate + iron(III) nitrate -> sodium nitrate + iron(III) carbonate
(a) ![]()
(b) It is not redox (all atoms retain their formal charges)
(ii) aluminum + nitric acid (hydrogen nitrate) -> aluminum nitrate + hydrogen gas
(a) ![]()
(b) It is redox because Al and H are changing charge states
(c) Oxidation: ![]() , reduction:
, reduction: ![]()
(d) Oxidizing agent: nitric acid; reducing agent: aluminum
(iii) aluminum + bromine -> aluminum bromide
(a) ![]()
(b) It is redox because Al and Br change charge states
(c) Oxidation: ![]() , reduction:
, reduction: ![]()
(d) Oxidizing agent: bromine; reducing agent: aluminum
3. Given the equation: ![]()
(a) How many moles of SO2 are produced from 6.68 moles of O2?
![]()
(b) How many grams of FeS2 are required to react with 0.750 moles O2?
![]()
(c) How many grams of SO2 can be prepared from 25.0 grams of O2?
![]()
(d) If 31.8 grams of SO2 were isolated in problem 3(c) above, what is the percent yield of SO2?
![]()
4. Given the reaction: ![]()
(a) What is the limiting reactant?
To determine which
reactant is limiting, we need to see if ![]() is greater or less than
is greater or less than ![]() :
:
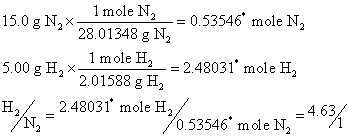
So ![]() is clearly greater
than
is clearly greater
than ![]() and so hydrogen is in
excess. That means N2 will go away first and stop the reaction which makes it the limiting reactant.
So we calculate the yield based on N2.
and so hydrogen is in
excess. That means N2 will go away first and stop the reaction which makes it the limiting reactant.
So we calculate the yield based on N2.
(b) What is the theoretical yield of ammonia (NH3) when 15.0 grams of N2 is mixed with 5.00 grams of H2?
![]()
5. Given the reaction: ![]()
(a) What is the theoretical yield when 20.0 grams of Mg are mixed with 10.0 grams of N2?
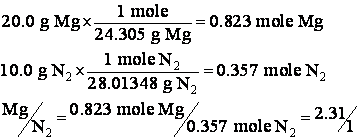
So ![]() is less than
is less than ![]() and so N2 is in excess and there isn't enough Mg. So Mg will go away first. We must
calculate the theoretical yield based on Mg.
and so N2 is in excess and there isn't enough Mg. So Mg will go away first. We must
calculate the theoretical yield based on Mg.
![]()
(b) What is the limiting reactant? Magnesium, because it goes away first, stopping the reaction.